How to Calculate Specific Heat Capacity
Use the dropdowns below to help you estimate what size air conditioner is best for your space. The SI unit of heat capacity is joule per kelvin JK.

Specific Heat Capacity Explained Youtube Business Plan Template Word Chemistry Lessons Letter Reversals
Specific heat of Sand is 830 Jg K.

. If your data facility has windows you must calculate how much heat is generated by sunlight from all the windows. Calculate the heat rate if steam enters a turbine at 500 degrees F and leaves at 300 degrees F at atmospheric pressure. BTUs per hour 12000 Tons per hour.
Specific heat tells you the amount of energy needed to raise each unit one degree. Heat Capacity is described in Joule per Kelvin JK. The molecules must vibrate in order to raise the temperature of the water due to the numerous connected hydrogen bonds.
For the same battery a discharge current of. For example a constant discharge current of 1 C 5 A can be drawn from a 5 Ah battery for 1 hour. Heat capacity or thermal capacity is a physical property of matter defined as the amount of heat to be supplied to an object to produce a unit change in its temperature.
C p C p M and. Specific Heat Capacity- Take equal quantities of water and oil at a temperature of about 30C. Know that specific heat refers to the energy needed to raise one gram by one degree.
Please note that in Heat capacity we consider the specific amount of mass and that mass can be any amount. C p c p. Using the energy equation of Q ṁ x Cp x ΔT we can calculate the cooling capacity.
The heat capacity problem can be applied to calculate the heat capacity mass or temperature difference of any given substance. Specifically it is a property of intensities. Metals - Specific Heats - Specific heat of commonly used metals like aluminum iron mercury and many more - imperial and SI units.
Heat capacity is an extensive propertyThe corresponding intensive property is the specific heat capacity found by dividing the heat capacity of an object. This is how specific heat capacity can be used to calculate the. Specific heat capacity.
The hydrogen bonding between water can be used to explain why water has such high specific heat. Weve rated and measured our units against a number of different room andor home conditions that can effect the cooling power required. Methanol with molecular formula CH3OH has a molar heat capacity C p of 811 Jmol K.
The calculated value does not match exactly but it is in the correct order of magnitude. This method is ideal for measuring the actual load of a process. How to Calculate How Many BTUs You Need.
A high heat capacity means that a substance can absorb a lot of heat before registering a change in temperaturethink about how long it takes for a pot to get warm to the touch on the stove versus how long it takes the water inside to get warm. ΔT the difference in degrees Fahrenheit. The specific heat capacity can be calculated from the molar heat capacity and vise versa.
Specific heat or specific heat capacity. Mass m 150 g. The presence of so many hydrogen bonds requires a greater amount of energy to break the water.
Specific Heat capacity of some substances at Atmospheric Temperature 20C. Ahmet Aktaş Yağmur Kirçiçek in Solar Hybrid Systems 2021. Specific heat C qmΔT 600 J150 g15C 0266 JgC.
W s In lbhr steam flow. Specific heat is a measure of heat capacity or how much heat a material can store when changing temperature. When you find the heat capacity of one unit of something 1 gram 1 ounce 1 kilogram etc youve found this objects specific heat.
C p molar heat capacity. Where R h The rate of heat in btuhr. Battery capacity is defined as the total amount of electricity generated due to electrochemical reactions in the battery and is expressed in ampere hours.
A good website for this is peacesoftwarede the although we will need to convert the units to imperial so for that we will use Specific heat capacity and density of water. To calculate a result you need to. A general calculation is 60 BTUhour per ft 2 of window but to get the exact numbers look at ASHRAEs specific formulas for these calculations for windows.
Heat 600 J. Poundshour X specific heat X ΔT BTUs per hour. Heat Capacity - The amount of heat required to change the temperature of a substance by one degree.
Calculate the rate of heat flux through a wall 3 m x 10 m in area A 30 m 2. The heat capacity of the substance is defined as the amount of heat required to raise the temperature of the substance by 1C. Specific Heat capacity in J kg 1 K 1.
Tons per hour x 12 safety factor Chiller size in tons. C btulb degree F specific heat capacity. M molar weight of the actual substance gmol.
Specific Heat Capacity of Water Explanation. Add a safety factor of 10 to 20. Calculate the specific heat of the substance.
C p specific heat capacity. Since they are difficult to calculate they are measured empirically and available in tables as references. This will give us a specifi heat capacity of 10007643BTUlbF and density of 62414lbFt3.
In thermodynamics specific heat capacity can be defined as the amount of heat needed to raise or lower the temperature of a unit mass of a substance by one degree Celsius. Calculate a simplified MCΔT. The cooling power of different types of air conditioners is based on BTUs.
Heating Up Applications - Energy Required and Heat Transfer Rates - Energy required to heat up a substance. Convert BTUs to tons. If youre given the amount of energy used the mass and initial temperature heres how to calculate the final temperature of a reaction.
The actual value for the specific heat capacity of water is 4200 JkgC. The wall is 15 cm thick L 1 and it is made of Sand with the thermal conductivity of k 1 025 WmK poor thermal insulator.

Specific Heat Capacity Amount Of Energy Needed To Change Temperature Of An Object By One Degr Business Plan Template Word Teaching Chemistry Chemistry Lessons

Specific Heat Capacity Problems Calculations Chemistry Tutorial Ca Learn Physics Chemistry Jokes Chemistry
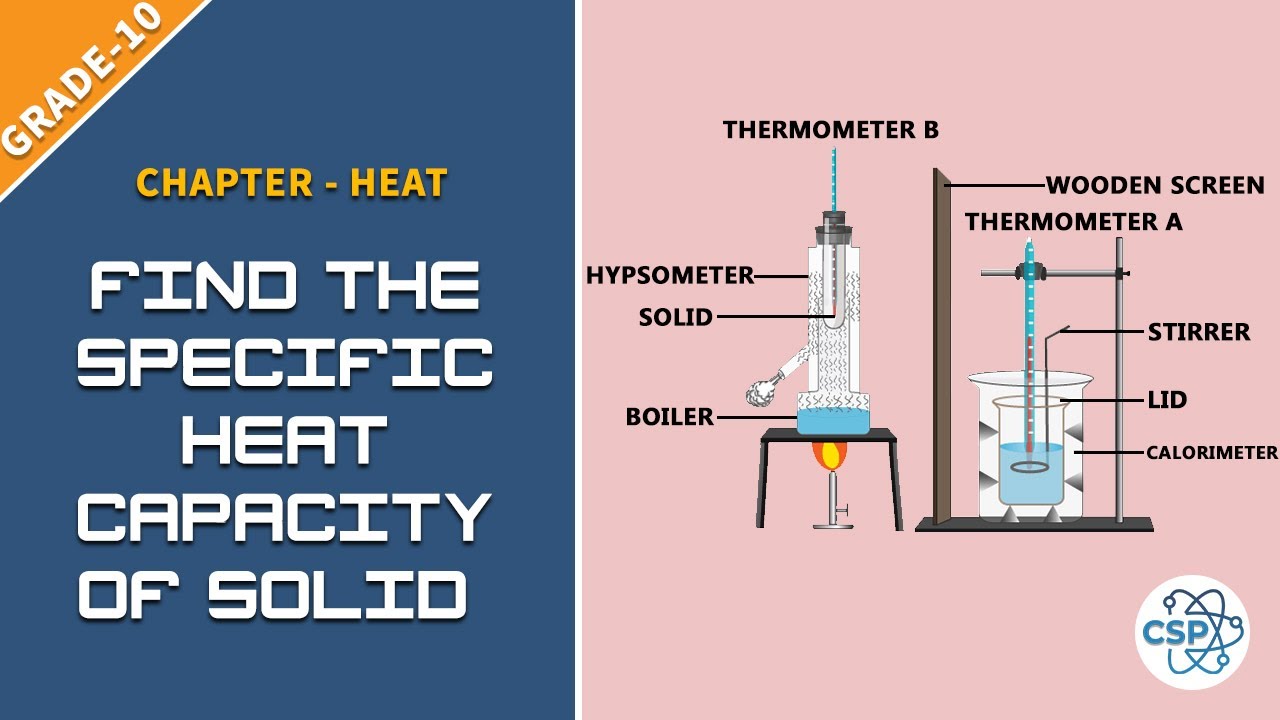
To Find The Specific Heat Capacity Of Solid By Using Method Of Mixtures See Class 10 Physical Properties Heat Science Experiments

No comments for "How to Calculate Specific Heat Capacity"
Post a Comment